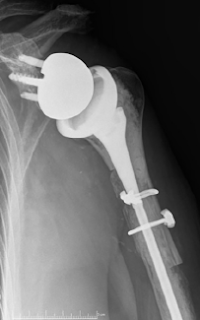
These authors reported their results for 73 patients treated with a reverse shoulder allograft–prosthetic composite (APC) because of proximal humeral deficiency.
On removal of the stem, serial broaches were used for successive trialing until stability was achieved. Bone loss was assessed at this stage by measuring the gap from the remaining native humeral bone distally along the medial cortex and the inferomedial portion of the polyethylene trial in the humeral component.
If the bone loss was deemed sufficient to compromise the stability of the revision prosthetic stem, then an allograft–prosthetic composite (APC) was chosen to provide enhanced stability of the construct. The fresh-frozen bulk humeral allograft was shaped using an oscillating saw and a step-cut technique. The prepared allograft was then cabled to the host bone using multiple 1.7-mm cables, and the definitive humeral component was cemented into the final construct. The remnant of the allograft subscapularis insertion was used to achieve repair to the remaining native subscapularis with multiple nonabsorbable sutures
The minimum follow-up period was 2 years, with an average of 67.9 months. 43 had more than 5 years’ follow-up and 12 had more than 10 years’ follow-up.
The Simple Shoulder Test score improved from 1.3 to 3.5 while the American Shoulder and Elbow Surgeons score improved from 33.8 to 51.4.
Good to excellent results were reported in 42 of 60 patients (70%), 10 patients (17%) reported satisfactory results, and 8 patients (13%) were unsatisfied. Range of motion improved in forward flexion (49° to 75°, P < .001) and abduction (45° to 72°, P < .001).
The Simple Shoulder Test score improved from 1.3 to 3.5 while the American Shoulder and Elbow Surgeons score improved from 33.8 to 51.4.
Good to excellent results were reported in 42 of 60 patients (70%), 10 patients (17%) reported satisfactory results, and 8 patients (13%) were unsatisfied. Range of motion improved in forward flexion (49° to 75°, P < .001) and abduction (45° to 72°, P < .001).
Complications occurred in 19 patients (26%). There were 8 periprosthetic fractures, 4 dislocations, 3 patients with humeral loosening, 2 glenosphere dissociations, and 2 infections. In total, 14 patients (19%) required revision. Of the revisions, 6 were for periprosthetic fracture; 2, instability with dislocation; 2, glenosphere dissociation; 2, humeral loosening; and 2, infection. Of the periprosthetic fractures, 3 occurred at the junction of the allograft and host bone and 3 occurred at the level of the distal tip of the humeral stem. Two of the distal fractures presented with radial nerve palsy after the fracture but before revision, and neither recovered at the time of most recent clinical follow-up.
There were 25 shoulders with type I bone loss (< 5 cm), 34 with type II bone loss (5 - 10 cm), and 14 with type III bone loss (> 10 cm). The rate of revision increased dramatically with the amount of bone loss. Revision was performed in 2 shoulders (8%) with type I bone loss versus 7 shoulders (21%) with type II and 5 shoulders (36%) with type III bone loss.
Revision was required in 14 patients (19%) for periprosthetic fracture (n = 6), instability (n = 2), glenosphere dissociation (n = 2), humeral loosening (n = 2), and infection (n = 2) at a mean of 38 months postoperatively. Ten patients had radiographic evidence of humeral loosening at final follow-up.
Comment: This series comes from the practice of a surgeon highly experienced in revision reverse arthroplasty. Surgeons with less experience may not have the same results. Even in these expert hands there was a substantial complication and revision rate, especially in those with massive bone loss.
The optimal allograft for this type of reconstruction is a size- and side-matched fresh frozen humeral allograft that can be stepcut to fit the remaining host bone. Because the number of such grafts is limited, surgeons should check on their availability before planning such a reconstruction.
The ability to obtain good fixation of the composite in distal host bone is important to the success of the procedure. Such fixation can be difficult to achieve if there is massive bone loss and if the canal size of the remaining host bone is small.
=====
How you can support progress in shoulder surgery
You may be interested in some of our most visited web pages arthritis, total shoulder, ream and run, reverse total shoulder, CTA arthroplasty, and rotator cuff surgery as well as the 'ream and run essentials'
=====
We have a new set of shoulder youtubes about the shoulder, check them out at this link.
Be sure to visit "Ream and Run - the state of the art" regarding this radically conservative approach to shoulder arthritis at this link and this link
Use the "Search" box to the right to find other topics of interest to you.
Be sure to visit "Ream and Run - the state of the art" regarding this radically conservative approach to shoulder arthritis at this link and this link
Use the "Search" box to the right to find other topics of interest to you.
How you can support progress in shoulder surgery
You may be interested in some of our most visited web pages arthritis, total shoulder, ream and run, reverse total shoulder, CTA arthroplasty, and rotator cuff surgery as well as the 'ream and run essentials'